Education
- Physics Engineering Syllabus By Ioe
Physics Syllabus For Engineering Entrance Examination In Nepal Given by Institute Of Engineering IOE :Unit I. Mechanics:DimensionsEquations of motion, Motion of a projectile. Laws of motion.Addition and subtraction of vectors. Relative velocity. Equilibrium...
- Millikan's Oil Drop Experiment
The oil drop experiment was an experiment performed by Robert A. Millikan and Harvey Fletcher in 1909 to measure the elementary electric charge (the charge of the electron).The experiment entailed...
- Discovery Of Electron By Discharge Tube Experiment
INTRODUCTIONGases are bad conductors of electricity. However under reduced pressure and at high potential difference gases conduct electric current.DISCHARGE TUBEDischarge tube is a glass tube fitted with two electrodes placed opposite to each other....
- Millikan, Robert Andrews (1868-1953)
US physicist, who was awarded the 1923 Nobel prize for Physics for his determination of the charge of a single electron and for validating Albert Einstein photoelectric equation. He also made important contribution to American science as an educator,...
- Discovery Of Ions
The person who gives a theory of ions is Michael Faraday. It’s around 1830. He describes the portions of molecules that move from anode to cathode or vice versa. He discovered that certain substances when dissolved in water conduct an electric current....
Education
MAIN PROPERTIES OF CATHODE RAYS
Do you know how electrons were discovered?If not click here 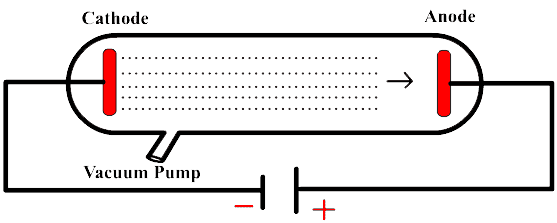 PROPERTIES OF CATHODE RAYS | ||
ORIGIN | ||
 These rays originate from cathode. These rays originate from cathode. | ||
PATH | ||
 Cathode rays travel in straight line. Cathode rays travel in straight line. | ||
PRESSURE | ||
 Cathode rays exert mechanical pressure. Cathode rays exert mechanical pressure. | ||
SHADOW FORMATION | ||
 The cathode rays consist of material particles because they produced shadow of objects placed in the way. The cathode rays consist of material particles because they produced shadow of objects placed in the way. | ||
EFFECT OF ELECTRIC FIELD | ||
 Cathode rays deflect in applied electric field towards the positive terminal. Cathode rays deflect in applied electric field towards the positive terminal. | ||
EFFECT OF MAGNETIC FIELD | ||
 Cathode rays are deflected by magnetic field. Cathode rays are deflected by magnetic field. | ||
PENETRATION POWER | ||
 Cathode rays penetrate small thickness of matter such as aluminum foil, gold foil, etc. Cathode rays penetrate small thickness of matter such as aluminum foil, gold foil, etc. | ||
CHARGE | ||
 These rays carry a negative charge. These rays carry a negative charge. | ||
DEPENDENCE ON MATERIAL | ||
 Cathode rays are independent of the material of electrode or the nature of gas in the tube. Cathode rays are independent of the material of electrode or the nature of gas in the tube. | ||
e/m RATIO | ||
 Their e/m ratio was equal to that of an electron. Their e/m ratio was equal to that of an electron. | ||
CONCLUSION | ||
| Different scientist tried different discharge tubes with different electrodes and different gases but results of all the experiment gave same value for charge to mass ratio. This shows that there is something common in all materials. It was concluded that all the substances in common. Experiment shows that these particles could be produces from any kind of material. Cathode rays consist of particles which are known as electrons. | ||
- Physics Engineering Syllabus By Ioe
Physics Syllabus For Engineering Entrance Examination In Nepal Given by Institute Of Engineering IOE :Unit I. Mechanics:DimensionsEquations of motion, Motion of a projectile. Laws of motion.Addition and subtraction of vectors. Relative velocity. Equilibrium...
- Millikan's Oil Drop Experiment
The oil drop experiment was an experiment performed by Robert A. Millikan and Harvey Fletcher in 1909 to measure the elementary electric charge (the charge of the electron).The experiment entailed...
- Discovery Of Electron By Discharge Tube Experiment
INTRODUCTIONGases are bad conductors of electricity. However under reduced pressure and at high potential difference gases conduct electric current.DISCHARGE TUBEDischarge tube is a glass tube fitted with two electrodes placed opposite to each other....
- Millikan, Robert Andrews (1868-1953)
US physicist, who was awarded the 1923 Nobel prize for Physics for his determination of the charge of a single electron and for validating Albert Einstein photoelectric equation. He also made important contribution to American science as an educator,...
- Discovery Of Ions
The person who gives a theory of ions is Michael Faraday. It’s around 1830. He describes the portions of molecules that move from anode to cathode or vice versa. He discovered that certain substances when dissolved in water conduct an electric current....
